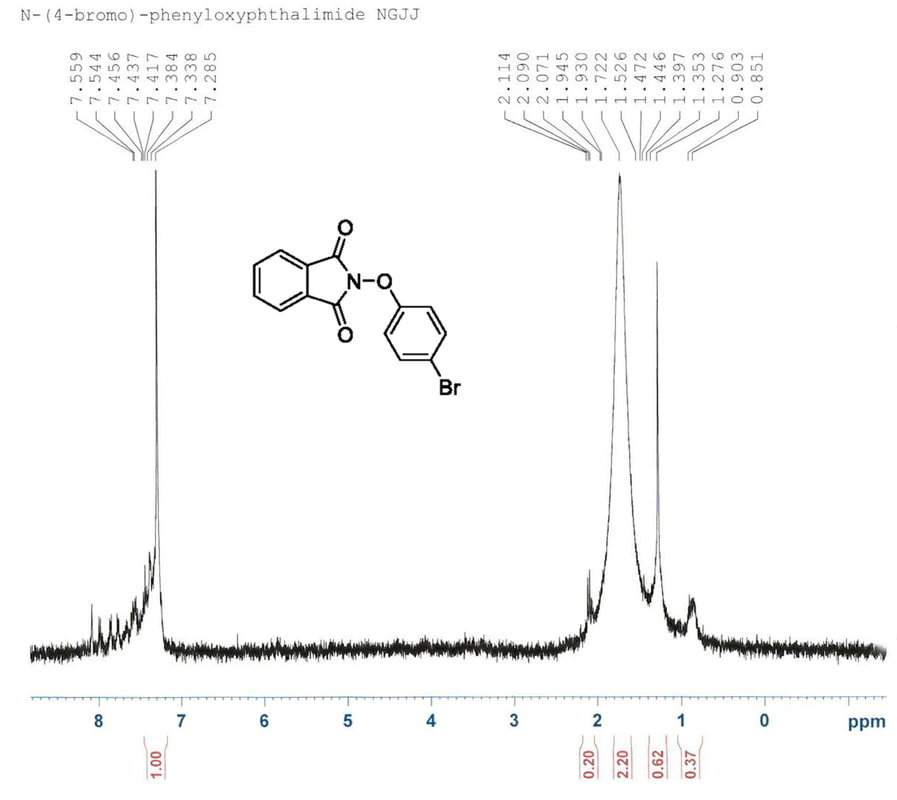
After research a bromine substituted N-phenyloxythalmide was successfully synthesized. According to DeSelm, the diarylodonium triflate salts created after step 1 are not stable and will not last one week in order to perform the second step. With this accounted for, in the initial attempt to synthesize N-phenyloxythalmide steps 1 and 2 were performed one day apart due to time constraints. The H-NMR taken after at this point showed obvious impurities, and peaks which were difficult to identify. It was evident that our method to create N-phenyloxyphthalimide was unsuccessful. The first NMR is shown below.
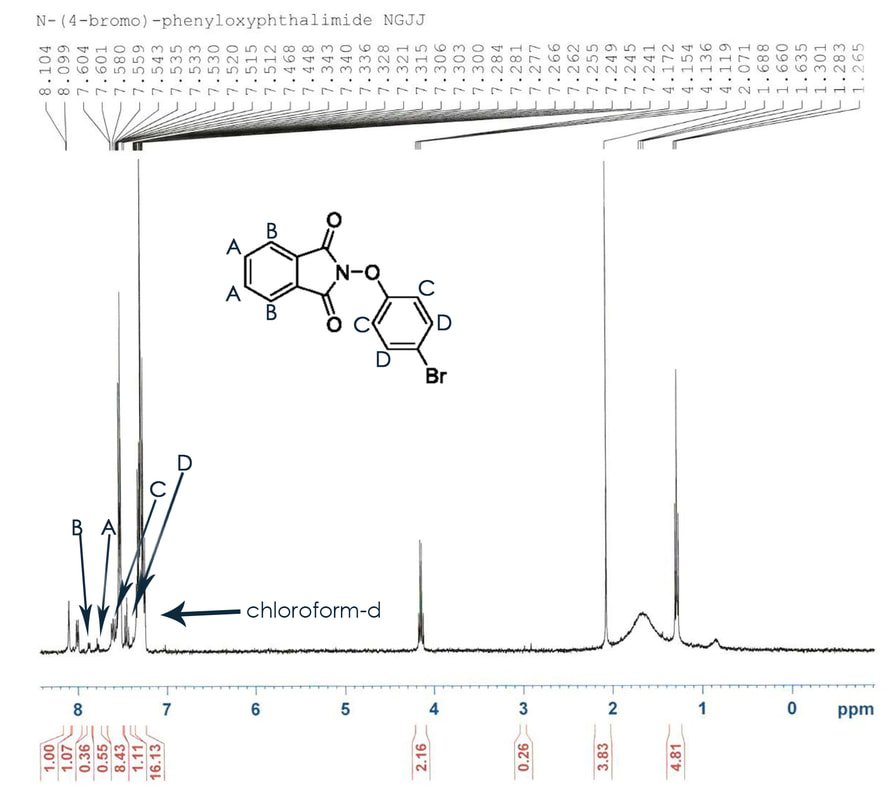
Although it was possible that the product sought after was present in the NMR it was clear that there were too many impurities in the compound necessitating another attempt. When executing the procedures the second time greater care was taken with measurements and temperatures and the first reaction took place immediately after the second. This H-NMR was supersaturated in hopes of amplifying the peaks that we sought after. Rather than dissolving a few crystals in chloroform-d, the chloroform-d was added directly to the solution. The H-NMR appeared much cleaner this time and is shown below.
The peaks between 7.4 and 6.8 are the peaks that resemble the N-phenyloxyphthalimide that was sought after step 2. Although the peaks are present, it is clear that there are many other impurities in the compound. In addition supersaturating the H-NMR sample did not have the amplifying effect that was expected as is clearly shown.
The compound was measured for mass and yield after the H-NMR. Since the chloroform-d was added directly to the compound, the compound was rotary evaporated to remove the chloroform. At this point the product we weighed at 0.346 mg and a 237% yield was calculated. Clearly this was incorrect so the compound was further inspected and it was discovered that the chloroform had not fully evaporator and the compound was rotatory evaporated once more. A second calculation revealed that the mass was actually 110mg and the percent yield was 75%.
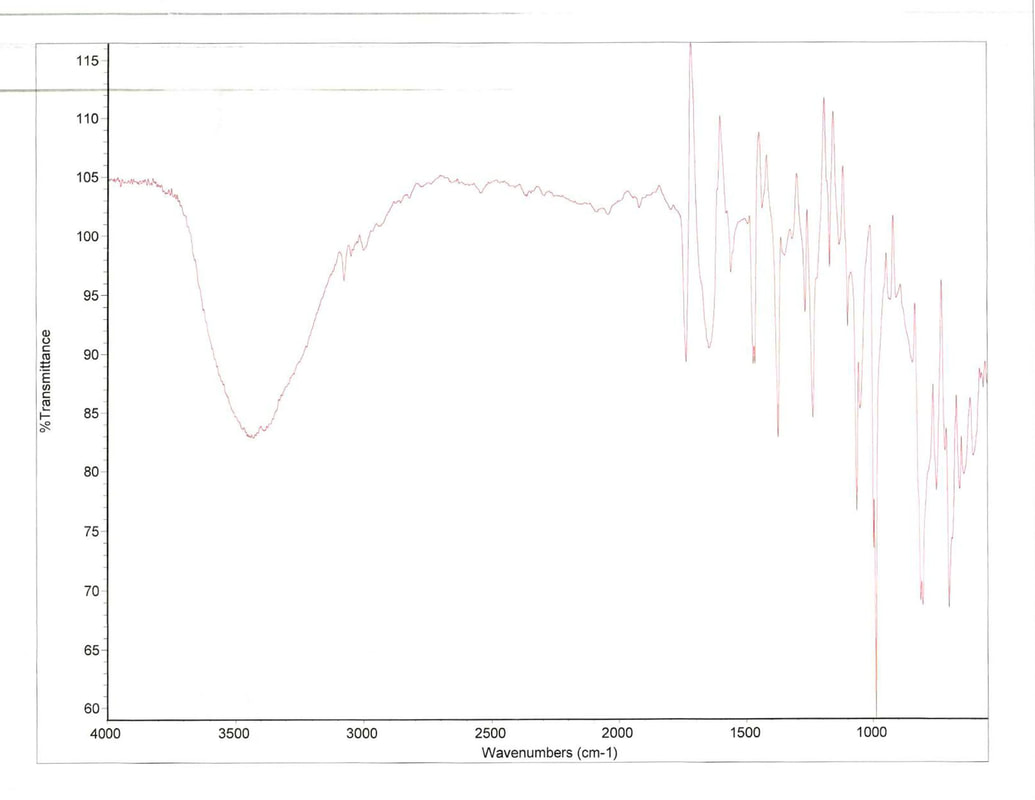
Another change was made during the second attempt of synthesizing N-phenyloxyphthalimide. During the first attempted it was noted that in step 2, upon adding brine to the solution to improve the separation, the solution turned cloudy and white indicating a precipitate had formed. Regardless a separation still occurred with a clear aqueous layer and cloud white organic layer. After the cloudy white layer was dried using anhydrous sodium sulfate, the solution was gravity filtered and whatever white solid had formed was removed. On the second attempt it was suspected that there could be some of the sought after product in this solid. Instead of filtering it out with the anhydrous sodium sulfate, a gravity filtration was performed first to remove the white solid. An IR scan was then performed of this solid to help quickly identify its importance. The scan is shown below.
The compound was measured for mass and yield after the H-NMR. Since the chloroform-d was added directly to the compound, the compound was rotary evaporated to remove the chloroform. At this point the product we weighed at 0.346 mg and a 237% yield was calculated. Clearly this was incorrect so the compound was further inspected and it was discovered that the chloroform had not fully evaporator and the compound was rotatory evaporated once more. A second calculation revealed that the mass was actually 110mg and the percent yield was 75%.
Another change was made during the second attempt of synthesizing N-phenyloxyphthalimide. During the first attempted it was noted that in step 2, upon adding brine to the solution to improve the separation, the solution turned cloudy and white indicating a precipitate had formed. Regardless a separation still occurred with a clear aqueous layer and cloud white organic layer. After the cloudy white layer was dried using anhydrous sodium sulfate, the solution was gravity filtered and whatever white solid had formed was removed. On the second attempt it was suspected that there could be some of the sought after product in this solid. Instead of filtering it out with the anhydrous sodium sulfate, a gravity filtration was performed first to remove the white solid. An IR scan was then performed of this solid to help quickly identify its importance. The scan is shown below.
The scan shows that the transmittance between 1700 and 3250 % are very low; it is in these region that we would find any aromatic, benzene rings, in our compound proving that the white substance could be disposed of. The procedures were continued as outlined from this point out.
Method III outlined by DeSelm will be executed for further research in attempt to successfully synthesize the novel anti-tumor drug. According to DeSelm method II that was outlined proved to be unsuccessful after several attempts and H-NMR scans.
In the future, assuming that the compound is successfully synthesized, O-(4-bromo)-phenyl-N-(9’-acridinyl)-hydroxylamine will have to go through many tests and trials. Further design will have to be developed to concentrate and purify the product. DNA binding analysis will be completed as done by Carlson in her research to identify the effects of the Bromine substituent group. In addition other derivatives with different effects on DNA, such as O-(4-methyl)-phenyl-N-(9’-acridinyl)-hydroxylamines, will be produced. After the products is thoroughly synthesized, purified, and analyzed it will go into testing before reaching the market.
Method III outlined by DeSelm will be executed for further research in attempt to successfully synthesize the novel anti-tumor drug. According to DeSelm method II that was outlined proved to be unsuccessful after several attempts and H-NMR scans.
In the future, assuming that the compound is successfully synthesized, O-(4-bromo)-phenyl-N-(9’-acridinyl)-hydroxylamine will have to go through many tests and trials. Further design will have to be developed to concentrate and purify the product. DNA binding analysis will be completed as done by Carlson in her research to identify the effects of the Bromine substituent group. In addition other derivatives with different effects on DNA, such as O-(4-methyl)-phenyl-N-(9’-acridinyl)-hydroxylamines, will be produced. After the products is thoroughly synthesized, purified, and analyzed it will go into testing before reaching the market.